There is a time and place for prescribed drugs, even those that have a high risk for dependence and misuse. For example, since cocaine has potent vasoconstrictor and anesthetic properties, it is used to suppress blood loss during corrective or cosmetic surgeries of the nose (MacNeil, et. al. 2020). Ritalin is used for treatment of ADHD. In this blog, I want to take a look at the process by which drug molecules are discovered, tested, and then approved for use by the Food and Drug Administration (FDA). Psychoactive drugs are then classified in a series of schedules by the Drug Enforcement Administration (DEA), according to the risk of dependence and misuse. The federal agency responsible for approving new drugs.
During the 19th century and the early part of the 20th century, the U.S. government did not engage in drug regulation in order to protect the public, nor did it require standards of efficacy and safety. In fact, it was possible for anyone to produce and market a concoction which presumably could cure any ill. More often than not, the concoction had significant amounts of alcohol and or opium, so of course the patient would feel “better”, but would not necessarily be cured. In the case of Jamaican Ginger, it was used as a remedy for headaches, upper respiratory infections, menstrual disorders, and intestinal gas…and it was 70-80% ethanol by weight.

By Deltabeignet (talk) – Self-taken photo. Label should be past copyright., Public Domain, https://commons.wikimedia.org/w/index.php?curid=133972026
This laissez-faire attitude changed with the election of Woodrow Wilson as president. Eventually, Congress passed legislation to create the Food and Drug Administration, or FDA, which would be charged with the responsibility of ensuring the efficacy and safety of therapeutic drugs.
The process of drug development
Before a drug is approved for public use, it must undergo a series of tests and stages of development which will provide sufficient information to the Food and Drug Administration to justify its use. In vitro and in vivo pharmacological effects form the rationale for considering a drug, and animal studies set the stage for clinical trials. The reason they are used is that they serve as models of human disease. The success of the model depends on how closely the pathophysiology in the animal model mimics that in humans. Animals can be used to investigate the relationship between drug dose and plasma concentration to beneficial and toxic effects. Furthermore, they can be used to screen for carcinogenic and teratogenic effects.
Human testing of drugs then progresses through a series of clinical trials. Phase I trials are usually single-blind, i.e. the health professionals know what is being administered to the patients, but the patients receiving the treatment do not know. The purpose of Phase I testing is to determine the maximum tolerated dose. All further dosing is done at concentrations less than the maximum determined with Phase I. Traditionally, healthy subjects were used. (For example, male prisoners were the first people to take female birth control pills.) More recently, patients for whom the treatment is intended are being used more frequently.
Phase II trials begin after the tolerated drug range has been defined. They are conducted to gather evidence that the drug has the effects which were suggested in preclinical animal trials. Tests are done to define the pharmacokinetics of a drug and to relate plasma concentrations to observed effects. Phase II trials may be simple, or double-blind, parallel or cross-over.
Phase III trials consist of definitive clinical trials that establish the efficacy and safety of the new drug. Whenever possible, trials are double-blind, randomized and controlled. They are almost always parallel in design.
Drug regulation and approval proceed by several steps. A pharmaceutical company will initially submit preclinical data to the FDA in a document called an Investigational New Drug (INA). The FDA will then give or withhold permission to initiate clinical trials in humans. The pharmaceutical company will then inform the FDA of progress, and when Phase III trials are completed, the company will submit all preclinical and clinical data to the FDA in a New Drug Application (NDA). The FDA reviews the data and decides whether they provide adequate documentation of safety and efficacy. Part of the approval process consists of writing a “label”, which includes the following information:
- Data that support approval;
- Pharmacological actions of drug;
- Indication (approved use) of the drug;
- A description of adverse effects;
- Instructions on dosing.
Drug development is a lengthy process. The time taken from submitting an application for approval to receiving a decision can take from 6 months to many years, although 1-2 years is typical. Therefore, the process of drug development, drug discovery to approval, takes from 6 to more than 10 years.
This lengthy and expensive protocol is supposed to protect the public from drugs which may lack efficacy, or produce dangerous or fatal side effects. Occasionally, there are cases in which drugs are withdrawn from the market because of unforeseen or unreported harmful side effects.
For example, on September 30, 2004, the Merck Pharmaceutical Company withdrew rofecoxib (Vioxx™) from the market after a doubling in the incidence of myocardial infarctions and strokes was documented among its users. With more than 80 million patients having taken this medication and annual sales exceeding $2.5 billion, this represents the largest prescription-drug withdrawal in history.
In an editorial appearing in the October 21, 2004 issue of the New England Journal of Medicine, Eric J. Topol, M.D., wrote that within a year and a half of the FDA granting approval to Merck to market rofecoxib, the Food and Drug Administration Arthritis Advisory Committee was aware of studies which showed an increase in the incidence of myocardial infarctions and strokes (Topol, 2004). He points out that the patients who are candidates for the pharmacological treatment of osteoarthritis also have established cardiovascular disease as well. As a result of Merck initiating an advertising campaign to dismiss concerns of cardiovascular safety, and of the FDA failing to demand a trial to specifically assess the cardiovascular risk and benefit of rofecoxib, tens of millions of patients taking rofecoxib were put at risk for major adverse events.
Reviewing drug safety and efficacy has also been conducted at the state level as well. In an article appearing on November 24, 2004 at CNN.com, “States take drug safety into own hands”, The Drug Effectiveness Review Project is described as providing member states (Alaska, Arkansas, Idaho, Kansas, Minnesota, Missouri, Montana, North Carolina, Oregon, Washington, Wisconsin and Wyoming) with information about specific drugs, so that the states can decide whether to pay for them. Although Merck & Co. withdrew Vioxx from the market in September 2004, John Santa, medical director for the Project, states that the data indicating that Vioxx was an unsafe drug were available years earlier.
The safety of other drugs approved by the FDA, including Meridia, Crestor, Accutane, Bextra and Serevent, have also been questioned in Congressional hearings convened in November, 2004, and this undermines the credibility of both drug manufacturers and the FDA.
This is obviously not the way the system is supposed to work in protecting the public. However, when a drug takes 8-10 years and costs $100-200 million to develop, the stage is set for selfish interests, enabled by complacency, to supersede consideration of the public good.
Psychoactive drugs which have the potential of abuse and addiction have to undergo a further step. The Drug Enforcement Administration (DEA) administers the regulations and governs the export of narcotic and non-narcotic substances. Controlled substances are listed in a series of schedules, as described below:
Psychoactive drugs are subject to scheduling as controlled substances
The Controlled Substances Act of 1996 empowers the Drug Enforcement Administration (DEA) to administer the regulations and to govern the export of narcotic and non-narcotic substances. Controlled substances are divided into 5 schedules, depending on their therapeutic application and on their abuse potential. These schedules include the following:
Schedule I Substances
- The drug or other substance has a high potential for abuse;
- The drug or other substance has no currently accepted medical use in treatment in the United States;
- There is a lack of accepted safety for the use of the drug or other substance other other medical supervision.
Some examples are heroin, marijuana, LSD, peyote, mescaline, psilocybin, tetrahydrocannabinols, ketobemidone, benzylmorphine, dihydromorphine, and morphine methylsulfonate.
The issue of marijuana
No one is arguing with the possible negative effects of marijuana use:
- Impairment of short-term learning and memory
- Possible paranoia or psychosis
- Possible hallucinations
- Association with amotivational syndrome
- Suppression of testosterone among males leading to hypermastia (enlargement of breasts)
At the same time, there are beneficial effects as well:
- Relaxation
- Appetite stimulation
- Heightened sensation
The placing of marijuana as a Schedule I controlled substance, which are defined as having no currently accepted medical use, high potential for abuse, and lack of accepted safety is problematic given recent changes in social attitudes and in state legislation. Dickson, et. al. (2019) report that among those states where marijuana is legal, sales reached $8 billion in 2017 and are projected to reach $24 billion by 2025. As of September 2019, 33 states and the District of Columbia have passed laws legalizing marijuana in some form. Furthermore, 11 states and the District of Columbia have legalized marijuana for recreational use.
While federal law places marijuana as a Schedule 1 substance, state laws are commonly divided into 4 groups: medical use, High-CBD/Low-THC only, decriminalization, and recreational legalization for adults 21 years old and up (Dickson, et. al., 2019).
CBD is a non-psychoactive component of marijuana that is found in Epidiolex, a CBD-based epilepsy drug
Surprisingly, a synthetic form of THC, dronabinol (AKA marinol) is a medication used to treat nausea and vomiting that is common among cancer patients undergoing chemotherapy, and is classified as a Schedule III drug.
Continuing to Schedules II through V
The labels of drugs in Schedules II through IV must contain the following warning: “Caution: Federal law prohibits the transfer of this drug to any person other than the patient for whom it was prescribed.” Furthermore, manufacturing quotas are placed on drugs classified in Schedules I and II (Taylor, 2002).
Schedule II Substances
- The drug or other substance has a high potential for abuse;
- The drug or other substance has a currently accepted medical use in treatment in the United States or a currently accepted medical use with severe restrictions;
- Abuse of the drug or other substance may lead to severe psychological or physical dependence.
Except in emergency situations, Schedule II drug can be dispensed only with a written prescription. Federal law does not permit refills on these prescriptions, so each time a patient needs a quantity of Schedule II drug, a written prescription is required.
Schedule II controlled substances consist of certain narcotic drugs, and drugs containing amphetamines or methamphetamines as the single active ingredient, or in combination with each other. Examples of Schedule II controlled substances are: opium, morphine, codeine hydromorphone (Dilaudid), methadone (Dolophine), meperdine (Demerol), cocaine, Oxycodone (Percodan), and straight amphetamines and methamphetamines. Also in Schedule II are methylphenidate (Ritalin), amobarbital, pentobarbital, secobarbital, phencyclidine, and methaqualone.
Prescriptions for Schedule III, IV, and V drugs cannot be refilled more than six months after the original prescription has been written (Taylor, 2002).
Schedule III Substances
- The drug or other substance has a potential for abuse less than the drugs or other substances in Schedules I or II;
- The drug or other substance has a currently accepted medical use in treatment in the United States;
- Abuse of the drug or other substance may lead to moderate or low physical dependence or high psychological dependence.
Schedule III drugs include limited quantities of certain narcotic drugs, and non-narcotic drugs such as: derivatives of barbituric acid except those that are listed in another Schedule, such as glutethimide (Doriden), methyprylon (Noludar), sulfondiethylmethane, sulfonmenthane, nalorphine, benzphetamine, mazindol, and paregoric.
Schedule IV Substances
- The drug or other substance has a low potential for abuse relative to the drugs or other substances in Schedule III;
- The drug or other substance has a currently accepted medical use in treatment in the United States;
- Abuse of the drug or other substance may lead to limited physical dependence or psychological dependence relative to the drugs or other substances in Schedule III.
Schedule IV drugs include barbital, phenobarbital, methylphenobarbital, chloral betaine (Beta Chlor), chloral hydrate, ethchlorvynol (Placidyl), ethinamate (Valmid), meprobamate (Equanil, Miltown), paraldehyde, pentaerythritol chloral (Petrichloral), fenfluramine, diethylpropion, and phentermine.
Schedule V Substances
- The drug or other substance has a low potential for abuse relative to the drugs or other substances in Schedule IV;
- The drug or other substance has a currently accepted medical use in treatment in the United States;
- Abuse of the drug or other substance may lead to limited physical dependence or psychological dependence relative to the drugs or other substances in Schedule IV.
Schedule V drugs and substances consist of preparations containing moderate, limited quantities of certain narcotic drugs generally for antitussive and antidiarrheal purposes, which may be distributed without a prescription order.
8.3. “Better living through chemistry”, or, tweaking the drug molecule to maximize its desired effects.
All therapeutic drug molecules have both desired primary effects and undesirable side effects, which will vary depending on the circumstances of the individual requiring them. For example, opiates are used quite extensively for the management of pain among cancer, amputee, and hospice patients. For each of these patients, an assessment of the type of pain being experienced (acute, chronic, intermittent, phantom, stump, joint, visceral) is of utmost importance in determining which drug is most suitable. Although opiate molecules extracted from the opium poppy have a similar structure, they can be altered to vary their binding characteristics to target receptors, and that, in turn, will affect their potency and their duration of action, as shown in the following list, where molecular diagrams are from the Merck pharmaceutical directory.
Codeine: Oral equianalgesic dose 240 mg, excreted in urine, duration 4-6 hours
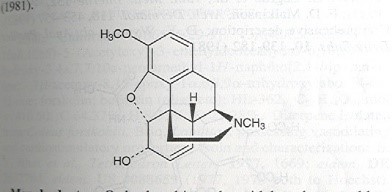
Hydromorphone: Oral equianalgesic dose 7.5 mg, excreted in urine, duration 4 hours
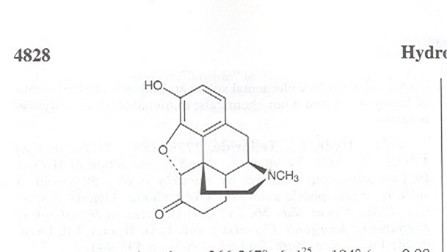
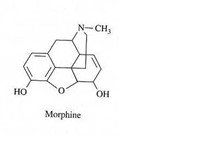
Morphine: Oral equianalgesic dose 30 mg, excreted in urine, duration 3-4 hours
Oxycodone: Oral equianalgesic dose 20 mg, excreted in urine, duration 4-6 hours
8.4. Many of the drug molecules discussed in this book are alkaloids.
Many of the drug molecules discussed in this book are alkaloids, a group of compounds which meet two descriptive criteria:
- The molecule has at least one nitrogen atom, derived from an amino acid; and
- The compound has limited distribution (Waterman, 1998).
Within this large group, we will find a veritable rogues’ gallery of neurotoxins, cardiotoxins, and psychedelics. Most of them are derived from higher plants and fungi, but a growing number of them are being discovered in algae and animals.
The consensus is that plants use them for self-defense, as a deterrent against herbivory. For most herbivores, the bitter taste of alkaloids is sufficient to make the herbivore cease nibbling or grazing on the plant . However, if the bitter taste does not suffice, the induction of nausea and vomiting, or the disruption of cognitive abilities by the herbivore will eventually force the herbivore to discontinue its eating habits.
Needless to say, humans have exploited the toxic nature of many alkaloids by extracting them and using them on poison arrows. In fact, Neuwinger’s contribution to Roberts’ and Wink’s Alkaloids: Biochemistry, Ecology, and Medicinal Applications is entitled “Alkaloids in Arrow Poisons” (1998). In that chapter, Neuwinger states “With a few exceptions, African and most Asian arrow poisons as cardiac poisons are absolutely deadly; there is no antidote, in contrast to the South American curare poisons, which normally can be survived with the use of antidotes or artificial respiration.”
Despite the toxicity of these compounds, medical science has extracted, or developed, an impressive list cornucopia of therapeutic drugs from alkaloids (Table 8.2).
| Table 8.2. Alkaloids used in modern medicine. Adapted from information in Schmeller and Wink (1998). | |
| Alkaloid name | Therapeutic Use(s) |
| Aconitine | Treatment of rhematism, neuralgia, sciatica |
| Ajmaline | Treatment of supraventricular and ventricular arrhythmias |
| Atropine | A) Antispasmodic in treatment of gastrointestinal, urinary and biliary colic; B) Preparatory for surgery; C) Used in combination with morphine to suppress its effect on vomitory center; D) Antidote for mushroom poisoning; E) Treatment for myasthenia gravis; F) Used in ophthalmology as a mydriatic and cycloplegic drug |
| Berberine | Antimicrobial |
| Boldine | Treatment of stomach and gastrointestinal disorders |
| Cocaine | Used as a local surface anesthetic and in corneal anesthesiology |
| Codeine | Antitussive, analgesic, mild sedative |
| Colchicine | Treatment of acute and recurrent gout |
| Emetine | Treatment of severe intestinal and hepatic amoebiasis; emetic |
| Ephedrine | Nasal decongestant; Treatment of edema in insulin-dependent diabetes; Bronchodilator |
| Ergometrine | Treatment of postpartum and postabortal hemorrhage |
| Ergotamine | Treatment of migraine headaches |
| Eserine = Physostigmine | Reduces intraocular pressure for treatment of glaucoma; Antidote for atropine poisoning |
| Galanthamine | Curtails muscle relaxant activity of tubocurarine and similar drugs |
| Hydrastine | Treatment of gastrointestinal disorders |
| Hyoscine = scopolamine | Prophylactic for motion sickness; Preparatory for surgery. |
| Lobeline | Treatment of bronchial asthma, chronic bronchitis, emphysema |
| Morphine | Analgesic; Treatment of diarrhea |
| Narceine | Treatment of cough |
| Papaverine | As a vasodilator, it is used to treat male impotence; Antispasmodic for gastrointestinal disorders. |
| Pilocarpine | Treatment of glaucoma and leprosy |
| Quinidine | Antimalarial; Treatment of ventricular and supraventricular arrhythmias. |
| Quinine | Treatment of malaria and babesiosis |
| Rescinnamine | Treatment of hypotension |
| Reserpine | Treatment of hypotension and chronic psychoses |
| Sanguinarine | Dental antiplaque agent |
| Sparteine | Treatment of cardiac arrhythmias |
| Strychnine | Treatment of eye disorders and optic nerve atrophy |
| Taxol | Antitumor agent for treatment of breast and ovarian carcinomas |
| Theobromine | Treatment of asthma and angina pectoris; Diuretic |
| Theophylline | Provides relief for bronchospasms due to asthma, bronchitis or emphysema |
| Tubocurarine | Induces muscle relaxation for surgical procedures |
| Vinblastine | Antitumor agent for treatment of Hodgkin’s disease and other lymphomas, testicular cancer |
| Vincamine | Vasodilator for cerebral disorders |
| Yohimbine | Aphrodisiac for impotent males; Treatment for urinary incontinence. |
Dickson, K.; Janasie, K.; Willett, K.L. (2019). A cannabinoid conundrum: A Study of Marijuana and Hemp Legality in the United States. Arizona Journal of Environmental Law Policy 10(20): 132-150.
MacNeil, S.D.; Rotenberg, B.; Soweby, L.; Allen, B.; Richard, L., Shariff, S.Z. (2020). Medical use of cocaine and perioperative morbidity following sinonasal surgery – A population study. PLOS One. https://doi.org/110.1371.journal.pone.0236356
Schmeller, T.; Wink, M. (1998). Utilization of Alkaloids in Modern Medicine. In Roberts, M.F.; Wink, M. (eds.) Alkaloids: Biochemistry, Ecology, and Medicinal Applications. Plenum Press, New York, PP. 397-433.
Taylor, W.N. (2002). Anabolic Steroids and the Athlete, 2nd edition. McFarland & Company, Inc., Publishers, Jefferson, N.C.
Topol, E.J. (2004). Failing the Public Health – Rofecoxib, Merck, and the FDA. New England Journal of Medicine 351:1707-1709.
Waterman, P.G. (1998). Chemical Taxonomy of Alkaloids. In Roberts, M.F.; Wink, M. (eds). Alkaloids: Biochemistry, Ecology, and Medicinal Applications. Plenum Press, New York, pp. 87-107.
